European Hematology Association Congress 2022
Building connections, pioneering therapies.
Agios Medical Booth
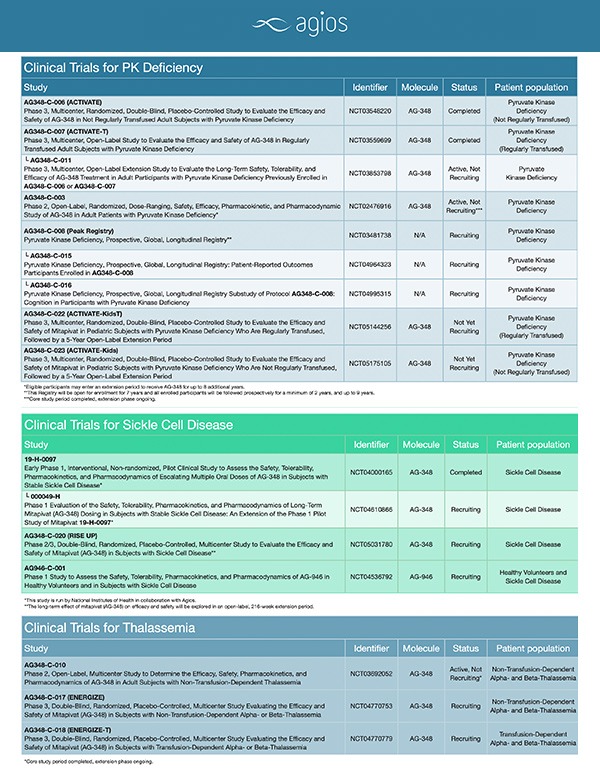
Clinical Trials
Agios is committed to developing innovative treatment options that make a meaningful difference in patients’ lives and fundamentally change the way genetically defined diseases are treated. Our leading clinical programs leverage our leadership in PK activation to explore investigational, oral treatment approaches for pyruvate kinase (PK) deficiency, thalassemia and sickle cell disease. More information about our clinical trials is listed below.
Thalassemia Clinical Program

ENERGIZE Phase 3 Trial
Learn more about our pivotal trial for non-transfusion dependent alpha- and beta-thalassemia.
Sickle Cell Disease Clinical Program
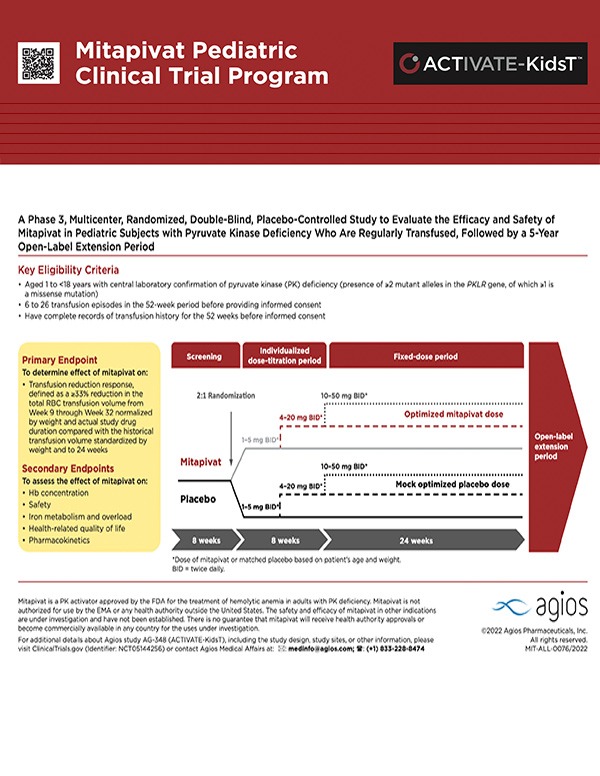
Pediatric PK Deficiency Clinical Program

ACTIVATE-Kids Phase 3 Trial
Learn more about our trial for pediatric non-transfusion dependent PK deficiency.
Peak Registry
Due to the rarity of PK deficiency, its prevalence, clinical burden, and long-term clinical course are not well defined. Peak is an ongoing, longitudinal, observational registry study designed to address this gap. Its primary objective is to record the natural history, treatment, outcomes, variability in clinical manifestations and disease burden of patients with PK deficiency. Approximately 500 patients are targeted for enrollment over 7 years at an estimated 60 study centers in up to 20 countries. Site and patient recruitment began in 2018 and all enrolled patients will be followed prospectively for at least 2 years (up to 9 years).
PK Deficiency White Paper
The white paper, entitled “PK Deficiency: Reflections on the Patient Experience to Support Treatment and Care,” shares the results of an international survey designed to explore communication between PK deficiency patients and caregivers and their hematologists. It explores in detail how the survey findings can inform approaches to improve PK deficiency disease management and patient experience. Given PK deficiency prevalence, the survey is thought to be the largest of its kind for the PK deficiency community to date.
The survey and white paper were developed by the PK Deficiency Advocacy Advisory Council (AAC), an international, multi-disciplinary group of experts, including patients, caregivers, patient advocates and clinicians, including representatives from the Thalassaemia International Federation (TIF), Metabolic Support UK, the National Organization for Rare Disorders (NORD), Thrive with Pyruvate Kinase Deficiency and the Pyruvate Kinase Deficiency Foundation. The vision of the AAC is that people around the world affected by PK deficiency receive timely diagnosis and can easily access the education, support and care they individually need. All AAC activities, including the survey and white paper, are funded by Agios.
Patient Resources
Podcasts for Patients
Agios is proud to be the sole sponsor of two podcast series designed to connect patients and caregivers with healthcare providers and innovators. We’re proud to introduce an all-new podcast focused on PK Deficiency. With hosts Dr. Zaidi and Dr. Callaghan now on the Agios team, we’re also advancing the next chapter of “CheatCodes: A Sickle Cell Podcast” while retaining its medical/scientific rigor and conversational nature.
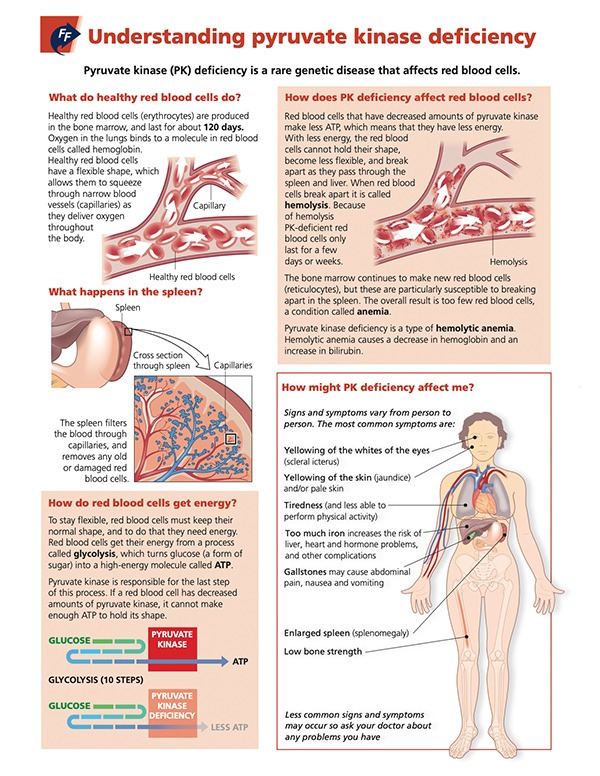
PK Deficiency Fast Facts
These patient-friendly resources provide more information about PK deficiency, symptoms and strategies for disease management.
More Patient Resources
View our list of vetted Agios and third-party resources for patients and caregivers. Resources are available for PK deficiency, thalassemia, sickle cell disease, metabolic and blood disorders, and rare diseases.
Explore Resources on Agios.com
Contact Us
Medical Information Inquiries
Agios Medical Information Call Center
Monday – Friday 8:00am – 6:00pm CT
Report an Adverse Event or Product Complaint Anytime
medinfo@agios.com
833-228-8474
MIT-ALL-0136